 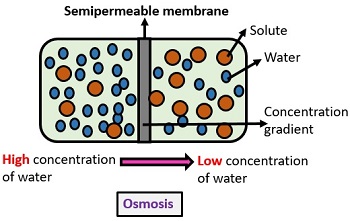
Diffusion is one of the processes that enable thi s. To enter and exit a cell, substances like water or nutrients have to pass through the semipermeable membrane. Dropping food coloring in a cup of water, which will change the color of the whole solvent (water), is another great example of diffusionĭiffusion is a widespread and important process for both nonliving and living systems. Spraying perfume in a room will make it smell nice for a little while, bu t over time d iffusion will distribute the odor molecules until their concentration is imperceptible to the human nose. Let’s look at some examples of diffusion in action. There is now an equal movement of chemical species in both directions. Once in equilibrium, the movement of molecules does not stop because their kinetic energy is the same. This effect is due to the higher kinetic energy of the hot water. Even though the food coloring was added to the hot water slightly after the cold water, it still diffused more thoroughly in this glass. The glass on the left contains hot water while the glass on the right is filled with colder water. If the concentration of a species is not initially uniform, over tim e d iffusion will cause a mass transfer in favor of a more uniform concentration.ĪDVERTISEMENT Yellow food coloring diffusing through water. Collisions between molecules are common : even i n the a ir at atmospheric pressu re, a molecule collides with a neighbor every few nanoseconds.Īcross the planet, the air inside the atmosphere has the same composition and is comprised of nitrogen (78%), oxygen (about 21%), argon (almost 1%), and other gases like CO 2 that are present in minute quantities (but still enough to warm the planet at an accelerating rate).ĭiffusion makes air composition uniform by re distributing chemical species, such as oxygen in the air, until equilibrium is reached : i n other words, until the concentration gradient - the difference in concentration between two areas - has been eliminated. The energy of the molecules causes random motion which in turn triggers diffusion. It is a measure of the average kinetic energy of the molecules in a material. Temperature, a physical quality people commonly reference in their daily lives, is directly related to molecular motion.  
Inside cells, diffusion is the transport of small molecules across the cell membrane. 
Credit: Comsol.ĭiffusion is the passive movement of molecules from an area of high concentration of the molecules to an area with a lower concentration. Over time, diffusion causes the solution to even up in concentration. Red is a high concentration of solvent while blue is the pure solute. ADVERTISEMENT What’s diffusion? Animation of a volume of solution which is initially nonuniform. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
